Sales of biologic medicines in Poland – including originators and biosimilars, outpatient and hospital ones – will exceed PLN 4.8bn (€1.1bn) in 2019. It will mark an impressive increase on 2018, a consequence of positive reimbursement developments, according to a new report from PMR, “Biologic and biosimilar medicines market in Poland 2019. Market analysis and development forecasts for 2019-2024”.
Good prospects for biologics market
In 2018, growth in the value of the Polish biologics market was held back by rapidly growing sales of biosimilars, which are cheaper than reference products. As a result, sales of several important drugs (active ingredients) declined in value. Originator biologics still dominate the Polish market in value terms: their development costs are much higher, and there are more of them than biosimilars. But the share of biosimilars has been growing, chiefly due to the emergence of biosimilar versions of several leading reference biologics that came off patent recently, such as Herceptin, Clexane, Humalog, or Humira. Between 2017 and mid-2019, the share of biosimilars in the Polish market for biologic medicines increased by five percentage points.
However, thanks to the inclusion of a relatively large number of reference biologics on the list of reimbursed medicines this year, the value of the Polish biologics market is poised to increase sharply in 2019. According to our forecasts, the market will grow by more than 20% compared to 2018.
Reimbursement key to market development
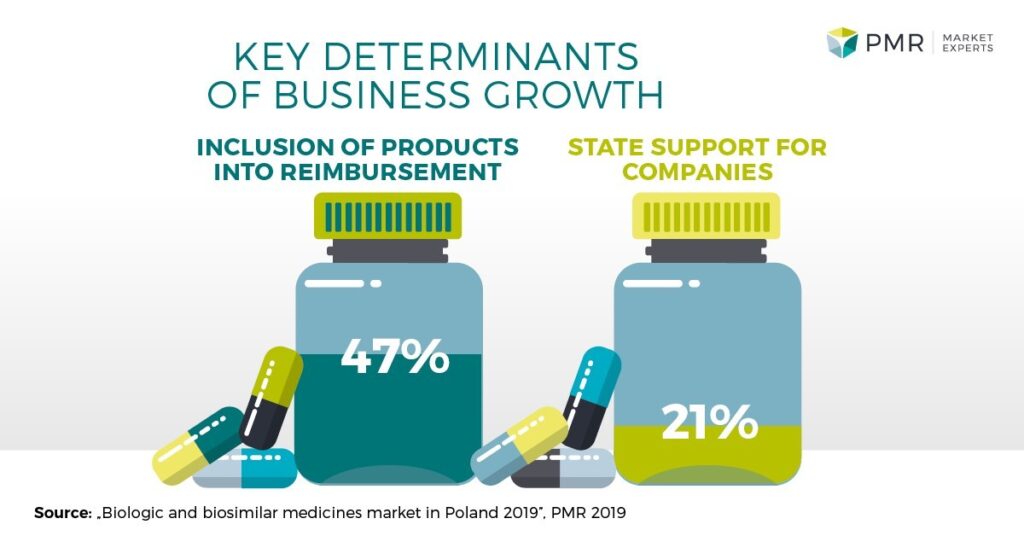
As part of the report, we conducted a poll of managers of companies active on the biologics market in Poland. One of the questions we put to our respondents was about key factors that supported or would support their business growth. The most indicated answer, mentioned by nearly half of the respondents, was inclusion of their products into reimbursement. This is understandable, given the prohibitive prices of most biologics. A fifth of the respondents would like the government to be more supportive of the sector, e.g. by undertaking public-private partnership (PPP) projects, instituting measures aimed at keeping intellectual property in the country (mentioned by the manager of a Polish firm), or providing support for early-phase clinical trials (mentioned by the manager of a Polish firm).
Other factors mentioned by the respondents included:
- educating patients and doctors about biologics and biosimilars
- growth of civilisation diseases
- adoption of a national programme for rare diseases
- a more predictable reimbursement policy.
AUTHOR
Monika Stefańczyk
Pharma & Healthcare Business Unit Director
MEDIA CONTACT
pr@pmrcorporate.com
Recent posts

PMR’s report: Poles cut down on expenses but fashion does not come first
PMR’s estimates indicate that in 2022 the market will record growth again, mainly on the back of increased demand for…

PMR: PLN 58bn online sales of services in 2022
Data from the latest PMR report “Online retail of services in Poland 2022. E-commerce market analysis and development forecasts for…

PMR: Record-breaking cosmetics market growth in 2022
PMR forecasts that in 2022 the cosmetics market in Poland will grow by 6.1% YoY, to around PLN 27.7bn. This…

PMR Hospital Outcomes Index highest for private hospitals
The situation on the hospital market over the last 10 years was at its worst in autumn 2020, according to…

PMR: Ready-to-eat meals market in Poland is stable in difficult times
The ready-to-eat meals market in Poland is growing rapidly, being in a relatively early phase of development. In 2021, this…

PMR: Value of pharmaceutical distribution in Poland reached PLN 56bn in 2021
According to the latest PMR report “Distribution on the pharmaceutical market in Poland 2022. Market analysis and development forecasts for…

